Supporting Your Global Market Success
Accelerated Mass Production for Faster Time-to-Market
⦁ Small-scale production & validation from DVT → PVT
⦁ Swift technology transfer
⦁ Cell Line for flexibility
End-to-End Production and Validation from DVT to PVT
⦁ Medical Device Technology Transfer and Validation
⦁ Production Process: Prototype Build → Pilot Run → Mass Production
⦁ Strict implementation of IOPQ process control, validation, and optimization ensures
stable mass production quality.
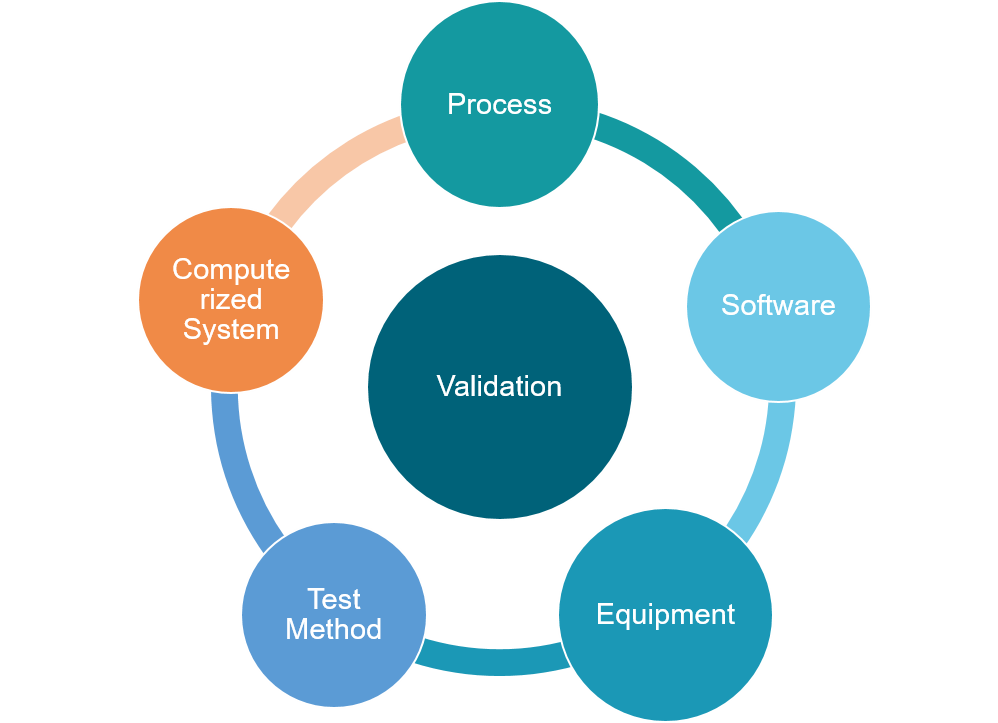
Comprehensive Process & System Validation
⦁ Complete validation of information systems, production processes, equipment, procedures, and software.
Tailored Assembly & Packaging Solutions
⦁ Flexible adjustment of assembly and packaging processes to meet diverse market needs and customer requirements.
Collaborative Design Partnership
⦁ Provides collaborative design and manufacturing services to enhance mass production feasibility, quality, and efficiency.
Global Market Support with Reliable and Consistent Quality
⦁ Capable of direct production and export of medical devices to major markets, including
the U.S., EU, and Japan.
⦁ One-stop development and mass production services, with a rapid understanding of
regulatory requirements to ensure global delivery quality and compliance.
Full Product Traceability
⦁ The Complete production history can be traced from the serial number (S/N).
One-Stop Medical Device Manufacturing Service
⦁ From product concept, regulatory identification, design, validation, pilot run,
clinical trial, certification, to mass production.
⦁ R&D innovation: leveraging group resources in hardware/software design, validation,
system integration, and production process improvement.
Commitment to Security
⦁ Comprehensive governance system covering organization, product design, and post-market
monitoring.
⦁ Established Product Security Incident Response Team (PSIRT) and Security Bounty
Program to strengthen product and service security continuously.
We ensure that every medical device undergoes comprehensive
validation to maintain integrity across the entire lifecycle.
No Matter the Stage of Development or Production, We Provide the Right Manufacturing Solutions

Support customers in applications or serve as a contract manufacturer:
Experience in major requirements:
